Description
Overview
XNano™ PCV is the world’s first fully enclosed, single-use LNP encapsulation
system designed for emerging applications such as personalized cancer vaccines,
in vivo CAR-T therapies, and more. It enables efficient production scaling from
40 mL to 4.5 L.
System setup is simple and can be completed in just 5 minutes
Preparation volume ranges from 40 mL to 4,500 mL
Designed with the QbD (Quality by Design) concept in mind, critical process
parameters are adjustable
Four-channel cartridge design accommodates a wider range of processes
Highly accurate flow rate control with real-time monitoring via an integrated
flow rate sensor
End-to-end system reduces the risk of contamination during both experimental
and production processes
Single-use fluid path eliminates the need for complex cleaning validation
Capabilities
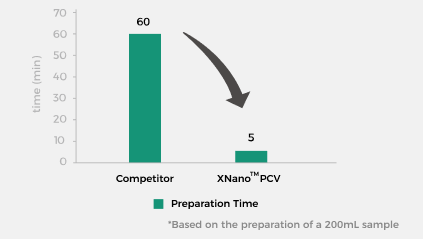
Simple and Efficient, No Cleaning Validation Required
The XNano™ PCV simplifies the process by eliminating complex steps such as
system cleaning. From assembly to formulation completion, the entire process
takes just 5 minutes*.
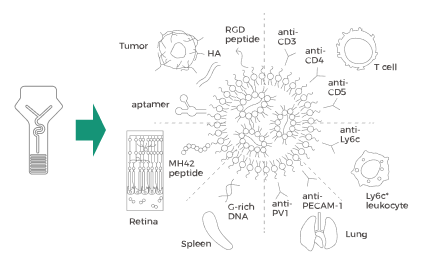
Innovative and Future-Focused
The patented four-channel cartridge design enables multiple dilution
processes while accommodating the requirements of differentiated delivery
systems. It expands application possibilities, including antibody-LNP
conjugation and core-shell delivery systems.
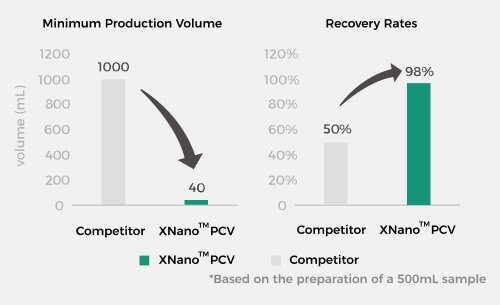
Cost Reduction and Efficiency Enhancement, Making Drugs Accessible
XGen's patented microfluidic technology and processes enable a minimum
preparation volume of 40 mL and a sample recovery rate as high as 98%*, making
drugs more accessible than ever.
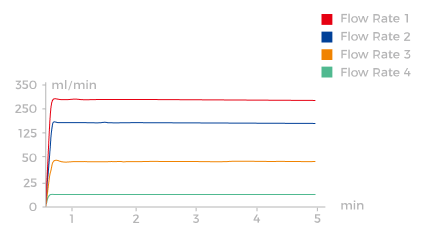
Intelligently Manufactured with 'Core' Precision
The I-Control and I-Sensor algorithms precisely control and display the
real-time flow rate with millisecond-level accuracy.
GMP Complicance
The equipment is meticulously designed and manufactured to comply with GMP,
cGMP, and EudraLex regulations. Adherence to ASME and BPE standards ensures
precision and quality throughout the process. Constructed with premium materials
and components, the equipment delivers exceptional stability and reliability,
meeting the highest industry standards.

Comprehensive Documentation and Full Traceability
The equipment's operating system is designed in strict compliance with FDA 21
CFR Part 11, incorporating multi-level user access management, data recording,
audit trails, electronic signatures, and may more additional features.







Reviews
There are no reviews yet.